|
For s-block and p-block elements, the valence electron is equal to the group number of the element. For calculating the valence electron we need to look at the groups in the periodic table. Any element located in groups 1, 2, 13, 14, 15, 16, 17, 18 will have the final digit of its group number as the number of valence electrons.Periods are horizontal rows of the periodic table, while groups are vertical columns (down) the table. Groups and periods are two ways of categorizing elements in the periodic table.Periodic table is a table listing the chemical elements in order of their atomic numbers.The alkali metal electron configurations. from the periodic table or from the electronic configuration. The periodic table can be broken into blocks that show what the last electron added to the electron configuration is. There are two ways to find valence electrons i.e.An electron that is part of an atom's outer shell and can take part in the formation of a chemical bond if the outer shell is not closed is known as a valence electron.JEE Main 2022 Question Paper Live Discussion.Difference Between Selling And Marketing.TS Grewal Solutions Class 11 Accountancy.Figure : The periodicity of valence electrons This table illustrates a number of interesting, and complicating, features of electron configuration. 
In groups 112, the group number matches the number of valence electrons in groups 1318, the units digit of the group number matches the number of valence electrons. Each element has a number of valence electrons equal to its group number on the Periodic Table. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. This is exactly how the periodic table is arranged, because the valence electrons go up the same way on the periodic table. TS Grewal Solutions Class 12 Accountancy The Periodic Table was designed with this feature in mind. 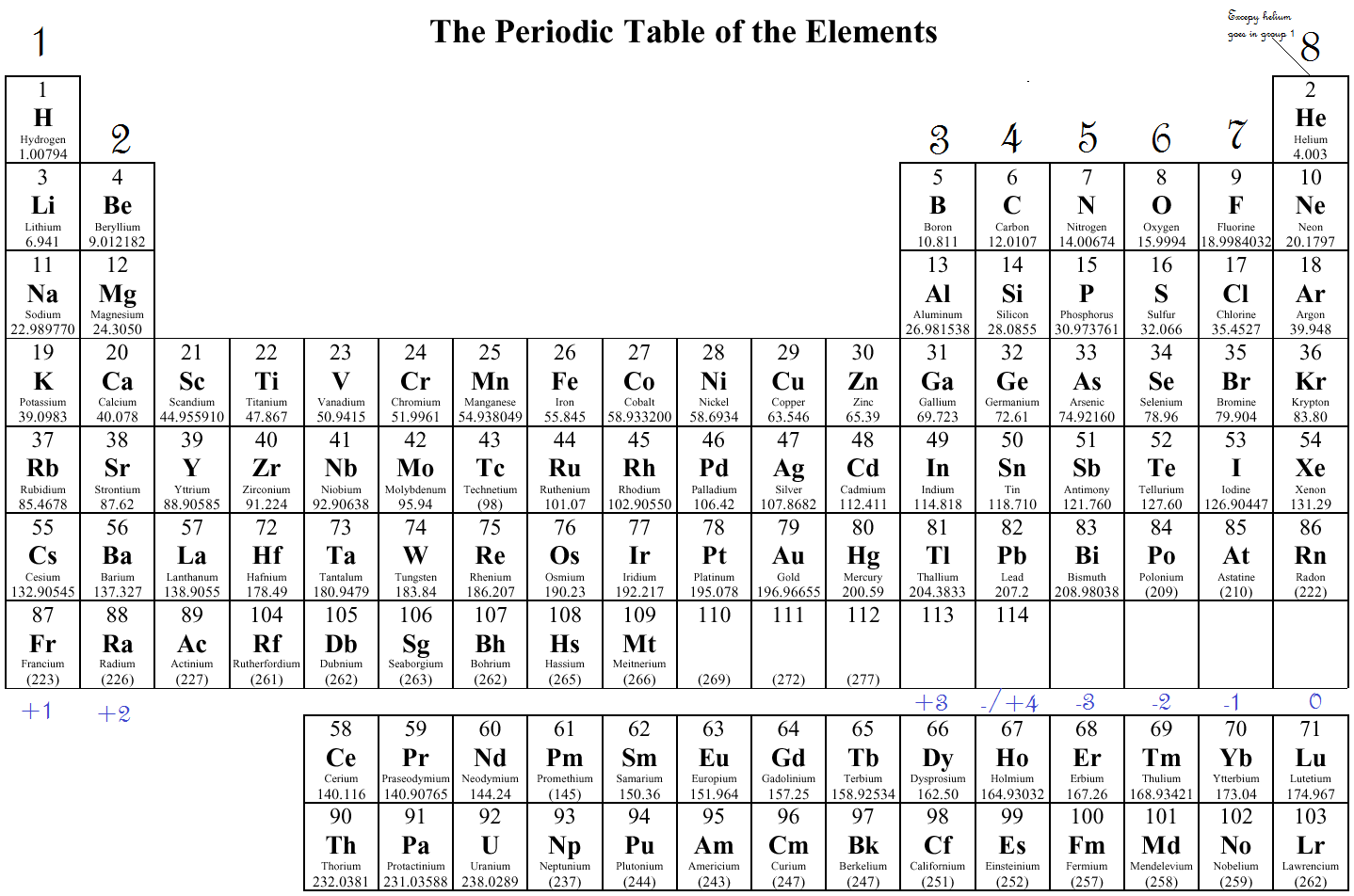
If we look at just the valence shell’s electron configuration, we find that in each column, the valence shell’s electron configuration is the same.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
